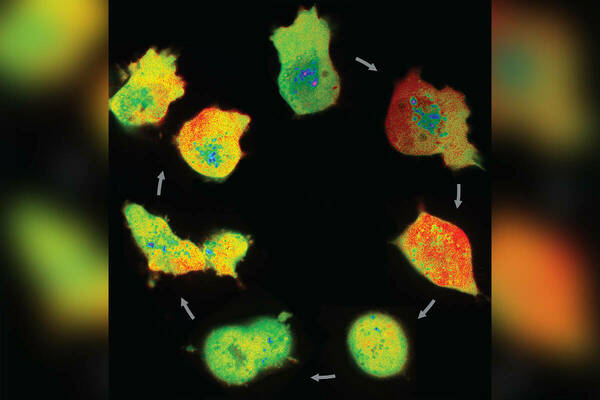
Scientists have known that the pH within cancer cells is generally higher than the pH within non-cancerous ones.

This increase in pH allows cancer cells to grow and divide more successfully, but previous studies were conducted using populations of thousands or tens of thousands of cells that could be in various stages of cell division.
A new study in the Journal of Cell Science by University of Notre Dame researchers examined individual cells instead, and showed that cancer cells have a higher pH than normal cells, but discovered that single-cell pH changes dynamically during the four phases of cell division. This discovery could eventually lead to therapies that will limit cancer cells’ ability to grow and divide.
Julia Spear, a doctoral student in the Integrated Biomedical Sciences program in the lab of Katharine White, the Clare Luce Booth Assistant Professor in the Department of Chemistry and Biochemistry, completed the research using normal and cancerous human lung cells and manipulating their pH to determine how they progressed through the cell cycle.
“Julia’s paper really adds on a significant understanding of what’s going on at the single- cell level,” said White, who is affiliated with Harper Cancer Research Institute. “There are big questions about where and when pH changes are happening during cell division and whether those changes are required or sufficient for cell division.”
[Watch as a single cell progresses through the cell cycle and divides into two daughter cells.]
White and Spear knew that pH dynamics within cells—intracellular pH (pHi)—signal them to start migrating or dividing. At the population level, previous research showed that preventing cells from having higher pH can delay division, but the mechanisms or reasons for this outcome were unclear.
Spear found that single-cell pH decreases when a cell decides to initiate the process of cell growth and division, but increases during the middle of the phase where each of the 23 chromosomes are duplicated. Later in that phase, however, pH decreases again, and increases just before the cell condenses its chromosomes for division. During the final phase of division, however, as the duplicated chromosomes separate into daughter cells, pH rapidly decreases again.

“Julia showed that increased pHi led to more successful division and that lowering pH in the cells really did suggest that if she was able to lower pH anywhere during the process, regardless of the actual raw value of pH, cells would have difficulty dividing,” White said.
Taken together, Spear’s data suggests lowering pH in cancer cells may block cancer cell division and growth. However, lowering pH of all cells in the body for cancer treatment simply is not an option, which has been a sticking point in targeting pH for cancer treatment.
“One of the issues is that most of those pH-lowering drugs fail in clinical trials because lowering pH damages very important tissues - your kidneys and heart,” White said. “This is why understanding the effects of increased pH is so important. It allows us to design drugs that target the underlying pH-dependent behavior.”
Understanding where the pH changes occur during cell division may someday help clinicians better treat the cells at the stage where lowering their pH would have the most benefit, Spear said.
Up next: The team is creating an experiment to find out which proteins sense the pH dynamics and shepherd the transitions between the phases of cell division, because that is one area the paper was not able to address.
Spear said she is excited to continue the fundamental research into the process of cancer cell division.
“While I’m not the one curing cancer, I’m showing the fundamentals of where and when pH changes might be important,” she said. “Then I’m giving someone else in the lab that data so they can identify the proteins to go after.”
The research was funded by an NIH Director’s New Innovator Award (DP2-DP2CA260416).
Originally published by at science.nd.edu on June 05, 2023.